 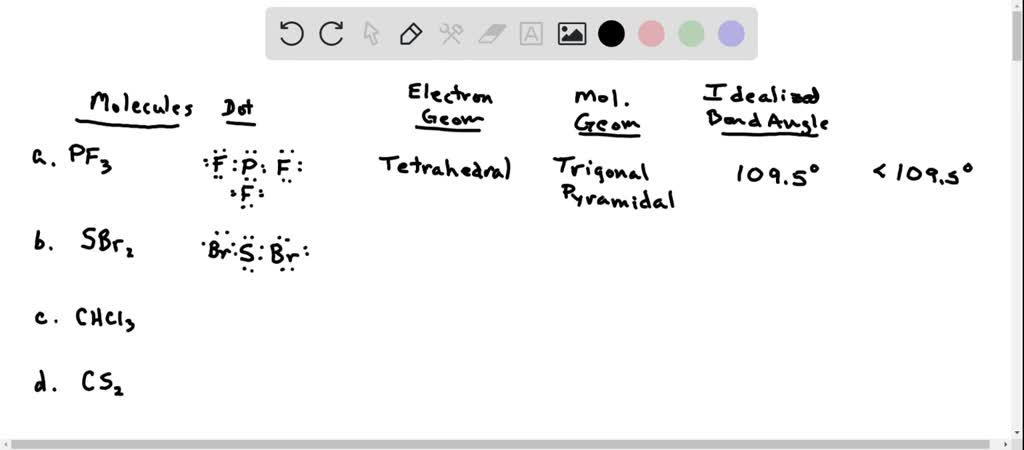
Hybridization of the given molecule H2S is sp3 the Sulfur atom is in center bonding with two Hydrogen atoms forming the bond angle less than 180 degrees. Now that we know the Lewis structure and hybridization of the molecule, it is easy to determine its molecular geometry. Hybrid orbitals and two lone pairs of electrons that make it an sp3 hybridization. In this case of H2S molecules, there are two sigma bonds, and there are two lone pairs of electrons on the central atom.Īs the Steric number of H2S is four, it has two = No of sigma bonds on the central atom +No of pi lone pairs on the central atom In this case, as Sulfur is the central atom the steric number of the compound will be the cumulative total of atoms bonded to the central atom and the number of lone pairs of electrons around the central atom. The steric number is defined as the region of electron density seen around the central atom.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
